
One Health Rationale to Investigate the emerGence of AMR related to chicken Meat and Egg consumption (OHRIGAME)

To understand the fate of antimicrobial drugs in poultry productions in South-East Asia (Vietnam) and the transboundary movement into the UK of hazards for human health (antimicrobial resistance and antimicrobial residues in meat).
Challenge

Global demand for animal protein is being met by intensification of livestock production especially chickens, where rapid industrialisation, particularly in Asia, is associated with increased use of antimicrobial drugs (AMD).
Overuse and misuse of AMDs in farming pose risks directly to local human populations due to:
- selective amplification of resistant (AMR) bacteria in animals that spill over to the local environment
- the presence in food of active antimicrobial residues which pose a direct chemical risk to people and/or promote selection and amplification of resistant bacteria in the human gut

To limit exposure to food-derived AMD, maximal residue limits (MRL usually in µg/kg) are defined and should not be exceeded in food destined for human consumption. These are international standards that may limit capacity of a country to export production in case of non-compliance.
In two rounds of field work (One Health Poultry Hub), we used mass spectrometry and found non-compliance in 9.2% of 141 meat samples on sale to the public in Vietnam (100x higher than the EU). This was due most of the time to failure to plan for the mandatory drug withdrawal periods before slaughter but sometimes the cause was unidentified and possibly related to occult entry to AMD in the production system.
The UK supply chain of chicken meat is vulnerable importation of hazards (AMR, residues) through international trade. Unintentional dietary intake though poultry products could pose a health risk to critical subpopulations of patients (e.g. liver transplant recipients) with increased incidences of gut dysbiosis and septic complications.
Solution
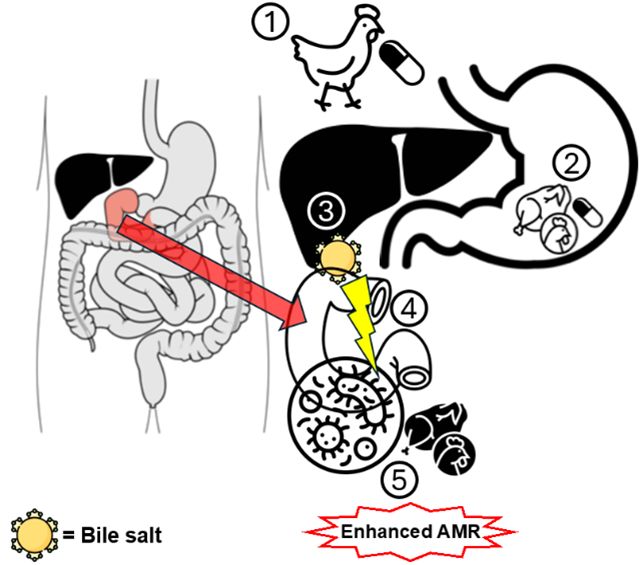
The OHRIGAME project aims to understand the fate of antimicrobial drugs in poultry productions in South-East Asia and the transboundary movement of hazards for human health (antimicrobial resistance AMR and antimicrobial residues in meat).
In Vietnam, this objective will be reached by:
- Establishing dynamics of AMR in gut microbiomes during production cycles, hypothesising high burdens of AMR in early life that are either conserved throughout the lifespan of the chicken or lost in time, depending on the nature of the antimicrobials.
- Identifying unaccounted sources of entry of AMD and AMR into production systems through occult sources. We will use forensic approaches to screen AMD in new matrices (feed, eggs, water).
- Understanding the social and economic contexts which may be associated or promote risky practices with regards to antimicrobial uses.

In the UK, this objective will be reached by:
- screening imported meat from South-East Asia at ports of entries and in retail samples for these hazards
- screening retail and hospital food for the presence of AMD residues
- Evaluating subclinical exposure to veterinary AMD and biliary microbiome of hospitalised patients admitted for a liver investigative or surgical procedures
Impact

Antimicrobial resistance is a truly global problem, and therefore it affects the whole world at human, animal and environmental levels.
OHRIGAME is a multidisciplinary project that integrates transnational studies from biological and social science disciplines and is designed to influence policies and practices in Vietnam.
The project in synergistic alignment with the new UK AMR National Action Plan for 2024 to 2029 and the Decision No. 414/QD-TTg to strengthen Vietnam Veterinary Service System capacity.
Astute AMR diplomacy actions executed by our UK-Vietnam collaborative team will support improved AMD stewardship in poultry production in Vietnam and assist in securing safe food supply for Vietnamese consumers. Additional impact include de-risking the import market from Vietnam, while ensuring level playing fields with regards to AMD use globally.
Partners
For further information on the human part of the project, see our sister page on the UCL website.







