FSTL3: A Crucial Regulator of Sertoli Cell Proliferation
Notwithstanding current world population, infertility in humans is on the rise. One in eight couples are infertile and in about a third of the cases, the defect arises from the male.
Testicular defects are among the principal causes of reproductive deficit in males. Perhaps due to environmental effects male infertility in animals is also on the rise. This has most likely contributed to the reduction in numbers of many animal species in the wild.
Understanding mechanisms that regulate testicular development and function is therefore essential for development of therapeutic strategies to alleviate male infertility disorders. The two major functions of the testis are to produce testosterone and sperm throughout the reproductive life of the species. Generally this begins at puberty when testicular size increases. Increased testicular size is an indicator of active generation of sperm and testicular function.
There is, however, a need for age-related waning of testis function so that mutations accumulated with age are not propagated in the population. For seasonal breeders an additional requirement is the cyclical increase and decrease of the testis with the beginning and end of each breeding season. Sertoli cells (SC) in the testis allow for the duplications and development of the cells that give rise to sperm and generally the total number of germ cells produced depends on the number of SC in a testis. This number is fixed very early in life and normally does not change with age. What is not clear is how the number of SC is determined and whether SC numbers in a testis can be increased later on in life. It is also unclear how testis size reduction is controlled in an age or season dependent manner.
We have found that a natural cell product, follistatin-like 3 (FSTL3) might be crucial in regulating testicular development. Our findings in genetically modified mice lacking FSTL3 in all cells show increased testis size, and a lack of testis size reduction with age. Also, within the testis there is an increase in SC numbers and related increase in cells that give rise to sperm. We, therefore, hypothesise that testicular FSTL3 action is essential for limiting testicular size and age-related decline in testicular function. There are only two other mouse models with increased testis size, but neither of these have a block in age-dependent reduction of testicular size. Our FSTL3 deletion mouse model, therefore, allows us to ask several crucial questions regarding testis development and function.
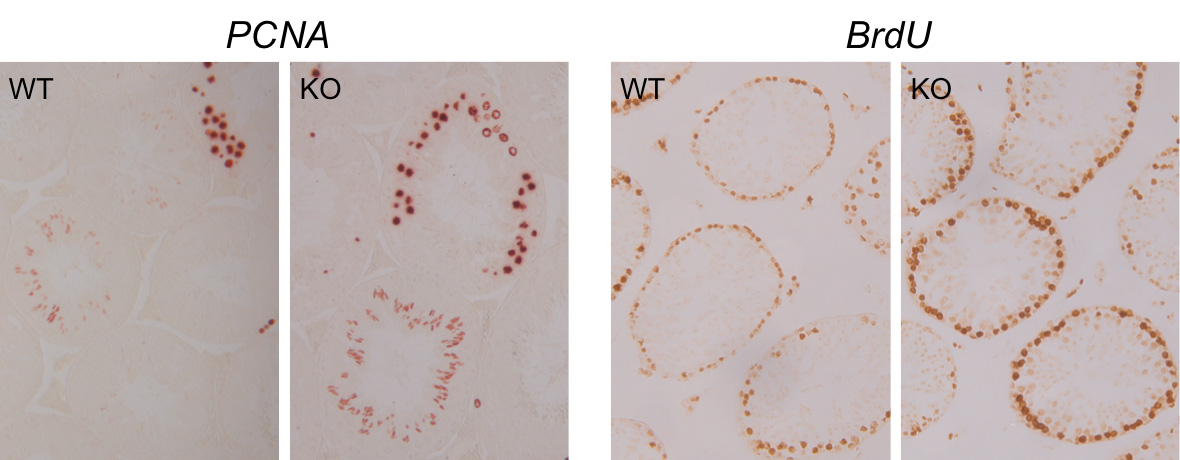
With a grant from BBSRC we will first determine whether SC multiplication can be increased by reducing FSTL3 in mice and in tissue culture cells. We will then investigate whether SC numbers, can be increased by deleting FSTL3 only in the SC. This will demonstrate whether SC numbers are controlled by local FSTL3 expressed in the SC or whether there is a systemic requirement for FSTL3 production. Furthermore, we will address whether removing FSTL3 after puberty increases SC numbers. This will demonstrate whether SCs can multiply beyond the first few days after birth, therefore providing the possibility of a therapeutic strategy of inducing SC growth and fertility in infertile males with limitingly small testes. Finally, we will identify which genes and cellular signalling pathways FSTL3 may influence to limit testicular size and help achieve reduction of testis size with age.
Our research will, therefore, identify how FSTL3 regulates testis development and function and helps limit reproduction with age. We will be identify the molecular mechanisms of these actions, whether FSTL3 dependent SC number regulation is achieved through its expression solely in SC and whether SC can be induced to grow, by removing FSTL3 at any time after birth. The contributions from this work to our understanding of the processes underlying testis development may support development of preventative and therapeutic approaches to dealing more effectively with male infertility.
